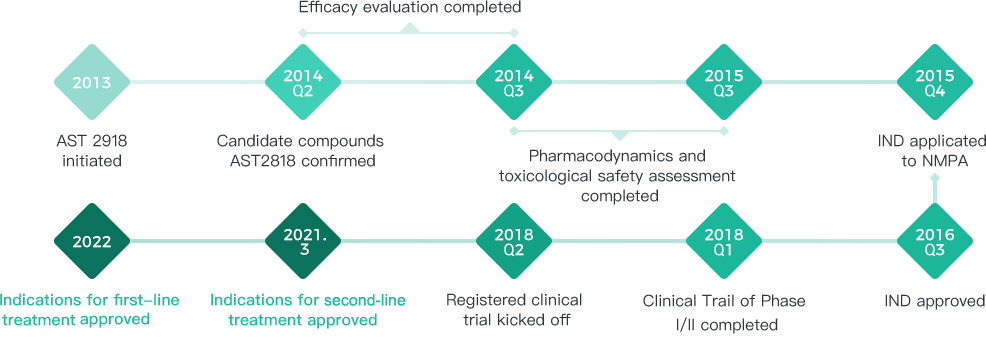
Pyridylamine pyrimidine derivative, preparation method and application of Furmonertinib obtained 17 patents in several countries


Furmonertinib
Furmonertinib is a Class 1 innovative drug developed by Allist that is intended to be used for the treatment of adult patients of locally advanced or metastatic non-small cell lung cancer (NSCLC) who experience progression during





 Drug instruction
Drug instruction